If a ruling by a federal judge in Texas is upheld, the decision has the potential to upend the nation’s abortion landscape. Here is what’s at stake and how the case may play out in the courts.
-
Send any friend a story
As a subscriber, you have “>10 gift articles to give each month. Anyone can read what you share.
Give this article
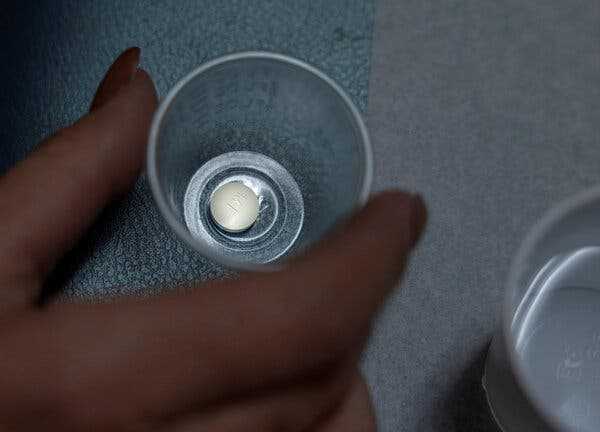
A federal judge in Texas invalidated the F.D.A.’s approval of an abortion pill, mifepristone.
The ruling by a federal judge in Texas last week revoking the Food and Drug Administration’s approval of the abortion pill mifepristone could be the most consequential abortion decision since the Supreme Court overturned Roe v. Wade in June.
But a lot remains uncertain. Here is what is at stake and what could happen next.
What’s at stake?
The ruling, by Judge Matthew J. Kacsmaryk of the U.S. District Court for the Northern District of Texas in a lawsuit filed by anti-abortion groups, invalidated the F.D.A.’s approval of mifepristone. That is the first pill used in the typical two-drug medication abortion regimen, which now accounts for more than half of the abortions in the United States. The plaintiffs claim that mifepristone is unsafe and that the F.D.A. did not adequately review the scientific evidence or follow proper protocols when it approved mifepristone in 2000 — claims that the F.D.A. strongly disputes, citing years of rigorous evidence that the drug is very safe.
If Judge Kacsmaryk’s ruling is upheld, the decision has the potential to upend the nation’s abortion landscape, making access more difficult in states where abortion is legal, not just states with abortion bans or restrictions. It could also have implications far beyond abortion, undermining the F.D.A.’s authority to approve and regulate other drugs and shaking the system that the pharmaceutical industry relies upon when it develops medicines.
Reproductive health providers and experts say that medication abortion, which is typically used in the first 12 weeks of pregnancy, would still be available in some form. Mifepristone might be sent from overseas, or the F.D.A. could decide not to take action against providers prescribing it. And some providers would switch to prescribing only the second abortion medication, misoprostol. But more patients would probably need to seek surgical abortions, and the ruling would create uncertainty and likely chaos throughout the country.
What happens next?
The first question is whether any court will grant the F.D.A. interim relief by extending Judge Kacsmaryk’s stay of his own decision, which is set to expire on Friday.
The Push to Restrict Abortion Pills
A federal judge in Texas invalidated the F.D.A.’s approval of an abortion pill, mifepristone. The decision could make it more difficult for patients to obtain abortions.
- Stockpiling Abortion Pills: As the ruling could affect availability even where abortion is legal, states led by Democrats have been scrambling to adjust to a possible future without mifepristone.
- Drug Companies React: The pharmaceutical industry issued a scorching condemnation in response to the ruling, calling for the decision to be reversed.
- Headwinds at the Supreme Court?: At first blush, the decision’s chances of surviving review by a Supreme Court dominated by conservative justices seem quite promising. But the justices may think twice before embracing it, legal scholars say.
Lawyers for the Biden administration have asked the U.S. Court of Appeals for the Fifth Circuit, in New Orleans, to rule on their stay application by noon on Thursday. To give itself more time to consider the question, the appeals court may grant an administrative, or interim, stay.
If that court ultimately grants an actual stay, the appeal will probably proceed in the usual fashion in the appeals court, with briefs and an oral argument in the coming weeks and months. The pill would continue to be available in the meantime.
At what point could it reach the Supreme Court?
There are several paths that could lead to Supreme Court review of the ruling.
It is possible that anti-abortion groups would ask the Supreme Court to lift a stay from the appeals court, but such an application would be an uphill fight. The drug has been on the market for 23 years, and an appeals court stay would restore the status quo. The justices would also know that they would almost certainly have an opportunity to weigh in later, after the appeals court rules on the merits.
The Fifth Circuit has a conservative reputation, and it may refuse to extend Judge Kacsmaryk’s stay or not rule on the government’s application, which would effectively be the same thing. The government would then ask the Supreme Court for a stay, probably on Thursday.
The government’s application would initially be addressed to Justice Samuel A. Alito Jr., the member of the court who oversees the Fifth Circuit. He would surely refer a matter of such consequence to the full court.
What might the Supreme Court do in the interim?
The Supreme Court may enter an administrative stay while it considers the application. Such stays do not necessarily predict whether the court will enter an actual stay.
If the court does enter a real stay, it has two basic choices about adjudication of the merits: to let the appeal proceed in the appeals court or to grant “certiorari before judgment,” leapfrogging the appeals court and proceeding to hear the case itself, which typically involves another round of briefing and oral arguments.
What happens if the appeals court acts first?
If the appeals court goes first, the justices would almost certainly afterward be presented with a petition from the losing party seeking review of the appeals court’s ruling. They would then have the opportunity, but not the obligation, to hear the case. Were the appeals court to rule in favor of the F.D.A., for instance, there is a fair prospect that the Supreme Court would deny review.
What if the Supreme Court takes the case?
If the Supreme Court takes the case right away, a procedure that has become more common in recent years, it could schedule an argument before the end of the current term, though that would be moving with unusual speed. Or it could set the case down for arguments in its next term, which starts in October.
Finally, it is possible that both courts will refuse to extend Judge Kacsmaryk’s stay. The case would continue, but the F.D.A.’s approval of the pill would be suspended, effectively deciding the merits of the dispute in at least the short term.
Why would a ruling in Texas affect the whole country?
Nationwide rulings from a single federal judge are not unusual, though scholars and some Supreme Court justices have expressed misgivings about them. The plaintiffs in the case, groups and doctors that oppose abortion, filed their lawsuit against the F.D.A. in Amarillo, Texas, where Judge Kacsmaryk, a Trump appointee who has written critically of Roe v. Wade, is the only judge responsible for that division on the court. Critics said that amounted to judge shopping.
Judge Kacsmaryk’s ruling faces competition from a contradictory order also issued on Friday by a federal judge in Washington State in another lawsuit related to mifepristone. That lawsuit, filed against the F.D.A. by Democratic attorneys general from 17 states and the District of Columbia, challenged extra restrictions that the agency imposes on mifepristone and asked the judge to prevent it from limiting access to the drug.
Judge Thomas O. Rice of the U.S. District Court for the Eastern District of Washington, an Obama appointee, did not lift the additional restrictions but ordered the F.D.A. to maintain the status quo. He blocked the agency from curbing the availability of mifepristone in the states that filed the lawsuit before that court, stopping short of entering a nationwide injunction. Still, those states make up the majority of states where abortion remains legal.
The Justice Department filed a motion on Monday asking Judge Rice to clarify what the F.D.A. would be obligated to do if the Texas ruling took effect — essentially seeking instructions from the judge that would allow the agency to continue to keep mifepristone available, at least while the Texas case works its way through the courts.
Source: nytimes.com



